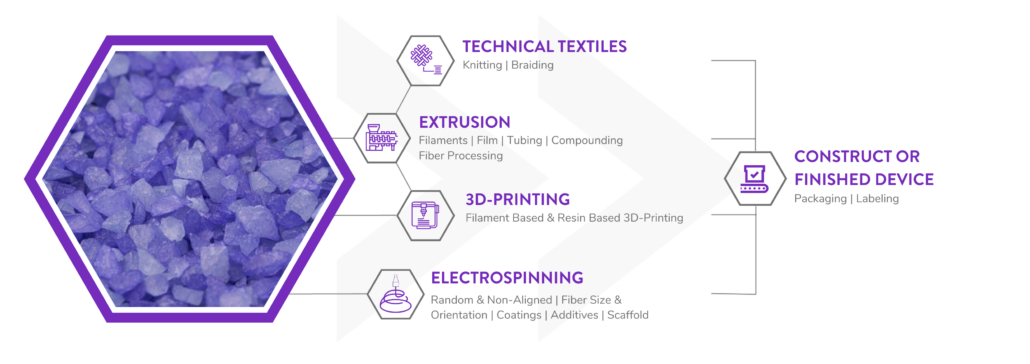
Advanced Absorbable Materials for Combination Medical Devices
Polymeric drug delivery systems have undergone significant development in the last two decades. Polymeric drug delivery has defined as a formulation or a device that enables the introduction of a therapeutic substance into the body. Biodegradable and resorbable polymers make this possible choice for lot of new drug delivery systems.
Challenges of drug administration:
Administration of a therapeutic requires optimization of dosing regimens to maximize therapeutic benefits while minimizing unwanted side effects to patients. This optimization is governed by four (4) basic pharmacokinetic principles: 1) absorption, 2) distribution, 3) metabolism, and 4) excretion. For any given active pharmaceutical ingredient (API), a number of considerations must be taken into account when designing dosage regimens which include: 1) routes of administrations, 2) site of therapeutic action, 3) the necessity of maintenance doses to ensure long-term therapeutic benefit, and 4) size of the therapeutic window.
Controlled release drug delivery systems:
Controlled release drug delivery systems have the potential to augment both the bioavailability and distribution profile of a given API. These systems have the ability to deliver APIs at a constant rate over long periods of time, resulting in decreased fluctuations in drug concentrations outside of a given APIs’ therapeutic window. In turn, this can decrease the dosing frequency for a given API and ultimately lead to increased patient compliance. The controlled release of APIs formulated for oral dosing is well-established through the use of complexation resins and coated reservoir systems. In contrast, non-oral controlled release drug delivery systems have historically been more challenging.
Parental drug delivery (e.g., intramuscular, intravenous, and subcutaneous administrations) is required for a number of APIs due to factors that include low bioavailability and a low tolerance for the chemical environments of the stomach and/or GI tract. Recent advances in excipient drug delivery techniques for parental administration have improved the ability to control a given API’s systemic delivery rate.
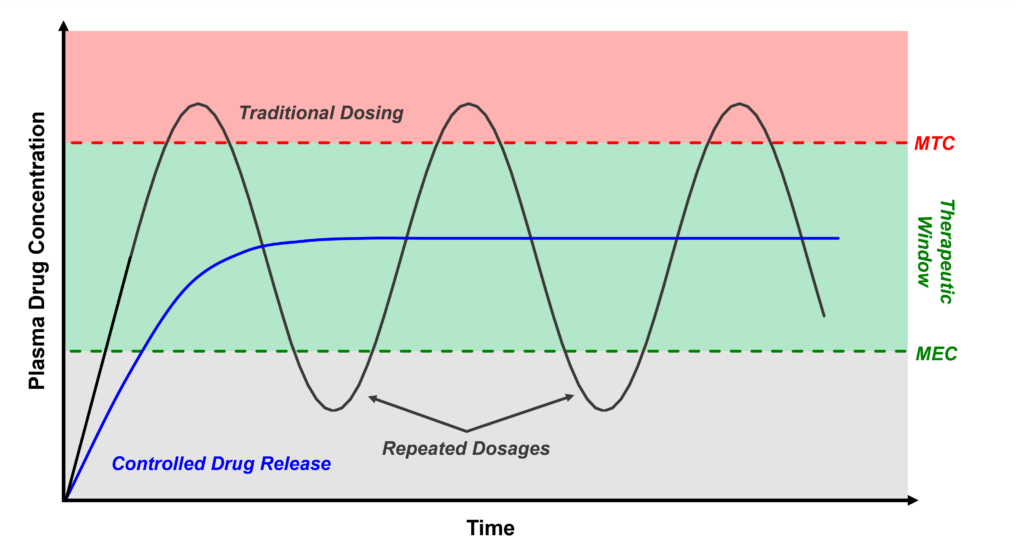
Electrospun materials:
Bioresorbable electrospun materials are an ideal excipient system given their ability to release APIs in a controlled manner over days rather than minutes/hours. These materials are also optimal due to their ability to act as a scaffold for large quantities and a variety of APIs while maintaining their biological activity. Bioresorbable electrospun materials can be degraded via natural metabolic processes and do not require surgical removal post-implantation.
Electrospinning is a process of manufacturing non-woven fibrous materials where a high voltage is applied to a probe connected to a polymer solution (which may contain an API). Once a sufficient amount of charge has accumulated to break the surface tension of the solution, a cone will form that allows for a liquid stream of polymer to be ejected at a continuous rate towards a spool. This material is then collected on the spool and can be used for downstream processing. Fibers produced via electrospinning exhibit diameters on the submicron scale (µm), causing these materials to have high surface area to volume ratios. Electrospun materials have successfully been produced for biomedical purposes ranging from controlled-release orally-dosed APIs, to wound healing applications.

Case study of successful preclinical long-term drug delivery strategy: Cisplatin
Various strategies have been developed for increasing the therapeutic benefits of certain APIs while decreasing their toxicity. Cisplatin is a highly toxic chemotherapeutic agent commonly used to treat a variety of different cancers. Cisplatin induces cell-death by causing non-specific DNA crosslinks to occur in not only cancer cells, but also healthy cells. This, in turn, causes both on-target regression of tumor cells, and several unwanted side effects that include nausea, kidney damage, nerve damage, heart failure, and hearing loss. IV administration of cisplatin results in high initial plasma drug levels which rapidly decrease with a half-life of roughly thirty (30) minutes. These plasma concentrations are contrasted with low tumor penetration, which limits the potential overall benefit of the therapeutic intervention. An ideal administration regimen of cisplatin would increase the tumor concentration of the API to maximize chemotherapeutic effects while simultaneously decreasing the plasma drug concentration to decrease unwanted side effects.
Preclinical studies by Shikanov, et al., 2011 illustrate this point. Using a bioresorbable polymer to control the delivery of cisplatin, the study was able to significantly improve therapeutic outcomes in a mouse model of bladder cancer while decreasing systemic exposure to the drug.
Remarkably, administration of cisplatin with the bioresorbable polymer directly into the tumor resulted in a five (5)-fold increase in the maximum tolerated dose (MTD) compared to systemic administration. In addition to increased tolerance of cisplatin, significant improvements in therapeutic outcomes and API distribution were observed. Local tumor injection of the bioresorbable excipient system resulted in 80% of subjects remaining disease-free for forty (40) days (i.e., the remainder of the study). This is in stark contrast to standard systemic administration of cisplatin which resulted in exponential tumor mass growth after seven (7) days. In line with the observed therapeutic benefits, local tumor administration of the bioresorbable excipient system resulted in drug levels at the site of administration over five-hundred (500) times higher than levels observed with systemic administration. Additionally, reverse trends were observed in systemic exposure where local tumor administration of the bioresorbable excipient system resulted in an eighty (80)-fold decrease in plasma drug levels compared to systemic administration. Together, this highlights the potential for polymer/API excipient systems to maximize exposure to the desired site of action, while simultaneously limiting systemic exposure to APIs and potentially decreasing side effects.
Absorbable polymers offered by Poly-Med, Inc. as excipients for drug delivery:
Poly-Med, Inc. (PMI), the leader in bioresorbable materials and medical device development, is vertically integrated with design, development, and manufacturing capabilities. PMI has a growing opportunity for providing materials for drug delivery via PMI’s unique Viscoprene® technology. Electrospun materials are generated using a combination of the Viscoprene® polymer that forms a base depot for drug delivery with additional polymeric diluents/APIs. Once combined, this drug delivery system can be injected through a standard Luer-Lok needle and syringe to be administered into the desired anatomical location. Additionally, PMI offers a catalog of Viscoprene® polymers, which allows for tailoring of the release properties of the API to meet the necessary clinical treatment schedule.
Poly-Med is a Vertically Integrated Bioresorbable Medical Device Manufacturing Partner:
Manufacturing bioabsorbable medical devices is hard. Controlling moisture levels and material degradation through the production cycle requires specialized equipment and process controls to ensure quality of finished devices. In contrast to other bioresorbable polymer manufacturers, Poly-Med produces polymer, is able to extrude this material to desired monofilament or multifilament formats, and is able process this material via warp knitting, weft knitting, or braiding processes to produce custom biomedical textiles. Beyond traditional textiles, we can process our bioresorbable polymers into non-woven formats via electrospinning via our state-of-the-art electrospinning facility. In addition to accessing Poly-Med’s more than thirty (30) years of experience manufacturing bioresorbable polymers, partnering with Poly-Med simplifies your bioresorbable medical device manufacturing supply chain. Contact us to today begin developing your custom bioresorbable medical device product line with a trusted partner for manufacturing advanced bioresorbable medical devices or purchase off-the-shelf high-performance bioresorbable polymers!